|
Dimethylglyoxime is a chemical compound described by the formula CH3C(NOH)C(NOH)CH3. Its abbreviation is dmgH2 for neutral form, and dmgH for anionic form, where H stands for hydrogen. This colourless solid is the dioxime derivative of the diketone butane-2,3-dione (also known as diacetyl). DmgH2 is used in the analysis of palladium or nickel. Its coordination complexes are of theoretical interest as models for enzymes and as catalysts. Many related ligands can be prepared from other diketones, e.g. benzil.
Preparation[edit]
Dimethylglyoxime can be prepared from butanone first by reaction with ethyl nitrite to give biacetyl monoxime. The second oxime is installed using sodium hydroxylamine monosulfonate:[1]
Complexes[edit]Ni Dmg 2 2 Structure 2
CID 83518 C8H14N4NiO4 CID 83518 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety.
Dimethylglyoxime is used to detect and quantify nickel, which forms the bright red complex nickel bis(dimethylglyoximate) (Ni(dmgH)2). The reaction was discovered by L. A. Chugaev in 1905.[2]
Cobalt complexes have also received much attention. In chloro(pyridine)cobaloxime[3] the macrocycle [dmgH]22− mimics the macrocyclic ligand found in vitamin B12.
Ni Dmg 2 2 Structure Games
Structure of chloro(pyridine)cobaloxime.
References[edit]
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Dimethylglyoxime&oldid=931825982'
Nickel bis(dimethylglyoximate) is the coordination complex with the formula Ni[ONC(CH3)C(CH3)NOH]2. The compound is a bright red solid. It achieved prominence for its use in the qualitative analysis of nickel.[1]
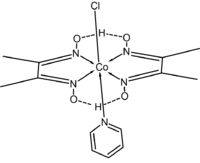 Structure[edit]Ni Dmg 2 2 Structures
Nickel(II) is square planar.[2] It is surrounded by two equivalents of the conjugate base (dmgH−) of dimethylglyoxime (dmgH2). The pair of organic ligands are joined through hydrogen bonds to give a macrocyclic ligand. The complex is distinctively colored and insoluble leading to its use as a chelating agent in the gravimetric analysis of nickel.
The use of dimethylglyoxime as a reagent to detect nickel was reported by L. A. Chugaev in 1905.[3]
Pressing Circle or B plants a mine that can be triggered multiple times by player or enemy attacks.MORE: Heavy BowgunThe heavy bowgun might seem redundant at first, but it fills a very different role than the light bowgun. Highest dmg weapons in mhw 2. Using all of the same controls and ammo types, heavy bowguns greatly decreaseyour mobility and the speed of your dodge roll, but increase your power-per-shot and rate of fire. You'll need to be quite good at predicting incoming attacks when using the heavy bowgun. Depending on what ammo you have loaded, you'll be able to deal damage from near and far. You'll also be able to dole out various elemental damage, stun monsters or even heal teammates.
Green gem cast when dmg taken poe 3. First off, what's your favourite CWDT combo? (supports etc) I'm still new to the game and would like to see how other people use cwdt Second, do you have any advice about my combo: if I'm running CWDT with immortal call AND molten shell, should I keep my CWDT level low so I get immortal call more frequently, even if it means keeping my gem levels low OR do I level everything up to get more out.
References[edit]
Ni Dmg 2 2 Structure 3
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Nickel_bis(dimethylglyoximate)&oldid=935161854'
Comments are closed.
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 RSS Feed
RSS Feed
